By
Ajala GLoria Eyitayo
All Saints University School of Medicine
Diego Gomez de la Garza
Universidad de Monterrey, Mexico
Amin H. Karim MD
Baylor College of Medicine
Houston, Texas
Author: Amin H. Karim MD
Recent Clinical Observer Group 2026
Students are given opportunity to study a case and write up a talk for the other students:





Students from Nigeria, Pakistan, Mexico, India, Nepal. 2026.

Observers from Turkey, Mexico, USA, Indonesia. April 2026
Peri-operative Cardiovascular Medication Management for Non-cardiac Surgery.

By Jessica Eve (Java, Indonesia)

REFERENCES
- Winchester DE, Mehta JJ, Alexander JT. Perioperative cardiovascular medication
management for noncardiac surgery. JAMA. Published online March 31, 2026.
doi:10.1001/jama.2026.0067 - American Heart Association. 2024 AHA/ACC guideline for perioperative cardiovascular
evaluation and management of patients undergoing noncardiac surgery. Circulation.
2024;150:e351–e442. doi:10.1161/CIR.0000000000001285 - Devereaux PJ, Mrkobrada M, Sessler DI, Leslie K, Alonso-Coello P, Kurz A, et al;
POISE-2 Investigators. Aspirin in patients undergoing noncardiac surgery. N Engl J Med.
2014;370(16):1494–1503. doi:10.1056/NEJMoa1401105 - Devereaux PJ, Yang H, Yusuf S, Guyatt G, Leslie K, Villar JC, et al; POISE Study
Group. Effects of extended-release metoprolol succinate in patients undergoing non-
cardiac surgery (POISE trial): a randomised controlled trial. Lancet.
2008;371(9627):1839–1847. doi:10.1016/S0140-6736(08)60601-7 - Neumann A, Maura G, Weill A, Alla F, Danchin N. Clinical events after discontinuation of
β-blockers in patients without heart failure optimally treated after acute myocardial
infarction: a cohort study on the French healthcare databases. Circ Cardiovasc Qual
Outcomes. 2018;11(4):e004356. doi:10.1161/CIRCOUTCOMES.117.004356 - Lindenauer PK, Pekow P, Wang K, et al. Lipid-lowering therapy and in-hospital
mortality following major noncardiac surgery. JAMA. 2004;291(17):2092-2099.
doi:10.1001/jama.291.17.2092 - London MJ, Schwartz GG, Hur K, Henderson WG. Association of perioperative
statin use with mortality and morbidity after major noncardiac surgery. JAMA Intern
Med. 2017;177(2):231-242. doi:10.1001/jamainternmed.2016.8005 - Berwanger O, de Barros E Silva PG, Barbosa RR, et al. Atorvastatin for high-
risk statin-naïve patients undergoing noncardiac surgery. Am Heart J. 2017;184:88- - doi:10.1016/j.ahj.2016.11.001
- Graham MM, Sessler DI, Parlow JL, et al. Aspirin in patients with previous
percutaneous coronary intervention undergoing noncardiac surgery. Ann Intern Med.
2018;168(4):237-244. doi:10.7326/M17-2341 - Kobori T, Onishi Y, Yoshida Y, et al. Association of glucagon-like peptide-1
receptor agonist treatment with gastric residue in an esophagogastroduodenoscopy.
J Diabetes Investig. 2023;14(6):767-773. doi:10.1111/jdi.14005 - Joshi GP, LaMasters T, Kindel TL. Preprocedure care of patients on glucagon-
like peptide-1 receptor agonists. Anesthesiology. 2024;141(6):1208-1209.
doi:10.1097/ALN.0000000000005231
Heart Failure With Preserved Ejection Fraction and Atrial Fibrillation: Egg or the Chicken
Heart Failure with Preserved Ejection Fraction and Atrial
Fibrillation: What was first, the Egg or the Chicken?
Laiba Ejaz
Karachi Institute of Medical Sciences.
Samuel Sotelo Hernandez
Autonomous University of Durango-Cappus Zacatecas
Amin H. Karim MD
Methodist Academy of Medicine, Houston, Texas
Abstract
A 68-year-old African American man presented with recurrent lower extremity edema without dyspnea or chest pain. His history included hypertension, hyperlipidemia, and obesity. Evaluation revealed atrial fibrillation with a
controlled ventricular rate and echocardiographic findings consistent with heart failure with preserved ejection fraction (HFpEF). Secondary causes such as deep venous thrombosis were excluded. The patient was managed
with metoprolol and diuretics for rate and volume control, losartan and hydrochlorothiazide for hypertension, apixaban for anticoagulation, and amiodarone for rhythm stabilisation. After a year of persistent atrial
fibrillation, elective cardioversion successfully restored sinus rhythm following confirmation of the absence of atrial thrombus on transesophageal echocardiography. The patient remained clinically stable on subsequent
follow-up visits.
Introduction
Atrial fibrillation (AF) and heart failure with preserved ejection fraction (HFpEF) are common cardiovascular conditions that frequently coexist, particularly in older, hypertensive, and obese patients. AF can exacerbate
HFpEF by impairing atrial contribution to ventricular filling, while HFpEF may predispose to atrial remodelling and subsequent arrhythmia, creating a complex bidirectional relationship. Both conditions independently
increase morbidity and mortality and pose challenges in diagnosis and management, especially when symptoms such as edema or fatigue are subtle or overlapping. Understanding the temporal relationship between AF and
HFpEF is critical for optimizing therapy, including rate versus rhythm control, anticoagulation, and management of comorbidities. This case highlights a patient with recurrent lower extremity edema, AF, and HFpEF, raising
the clinical question of which condition preceded the other and illustrating the importance of individualized, comprehensive cardiovascular care.
Case Presentation
A 68-year-old African American man presented to the clinic with complaints of recurrent fluid buildup, mainly in his lower extremities, as noted by his primary care physician. He denied dyspnea, chest pain, orthopnea, or
paroxysmal nocturnal dyspnea. His medical history included hypertension, hyperlipidemia, and obesity. He reported occasional alcohol use and denied smoking. His family history was significant for coronary artery
disease, hypertension, hyperlipidemia, and diabetes mellitus. On examination, his weight was 280 lbs (127 kg) and height 73 inches, yielding a BMI of 37.7 kg/m2, consistent with Class I obesity. Blood pressure ranged
between 135–145/75–85 mmHg, and his pulse was irregular at 58–70 beats per minute.
Laboratory
investigations revealed a normal HbA1c and a lipid profile with total cholesterol 222 mg/dL, HDL 67 mg/dL, LDL 149 mg/dL, and triglycerides 60 mg/dL. An electrocardiogram (EKG) (figure 1) obtained during the initial
visit showed atrial fibrillation with a controlled ventricular rate of approximately 58 beats per minute. A subsequent 2D echocardiogram demonstrated a preserved left ventricular ejection fraction of 60–65% and mild
left atrial enlargement—findings consistent with heart failure with preserved ejection fraction (HFpEF). Chest X-ray showed chronic airway changes and mild atherosclerosis, while Doppler ultrasound ruled out deep
venous thrombosis. The patient was started on metoprolol and diuretics for rate and volume control, along with losartan and hydrochlorothiazide for blood pressure management. Anticoagulation with Eliquis (apixaban) and
rhythm control with amiodarone was initiated and continued for over a year without spontaneous conversion to sinus rhythm (figure 2). He subsequently underwent elective cardioversion, which successfully restored sinus
rhythm (figure 3) A transesophageal echocardiogram prior to the procedure confirmed the absence of thrombus in the left atrium or left atrial appendage. The patient continued regular follow-up and remained clinically stable
thereafter.
Discussion
This case highlights the complex interplay between atrial fibrillation (AF), hypertension, obesity, and heart failure with preserved ejection fraction (HFpEF) in an older African American male. The patient’s presentation
with peripheral edema, in the absence of dyspnea or orthopnea, emphasises that HFpEF can manifest subtly and that careful evaluation of cardiovascular risk factors and cardiac rhythm is essential for accurate diagnosis and
management (1,2). Atrial fibrillation is a common arrhythmia in elderly individuals, often associated with structural heart changes such as left atrial enlargement and diastolic dysfunction. In this patient, chronic
hypertension and obesity likely contributed to increased left ventricular stiffness and impaired relaxation, leading to diastolic dysfunction and, consequently, HFpEF (3,4). The echocardiographic findings of preserved left
ventricular ejection fraction (60–65%) with mild left atrial enlargement are consistent with this diagnosis. The irregularly irregular rhythm on electrocardiogram confirmed the presence of AF, while the controlled ventricular rate suggested adequate rate control with beta-blocker therapy (5).
HFpEF accounts for nearly half of all heart failure cases, and its prevalence continues to rise in parallel with obesity, hypertension, and metabolic syndrome (1,6). Pathophysiologically , the combination of increased
ventricular wall stress, endothelial dysfunction, and myocardial fibrosis results in elevated filling pressures despite normal systolic function (7). This leads to symptoms such as lower extremity edema, exercise intolerance, or mild dyspnea, which may fluctuate depending on volume status. Importantly, obesity is a key modifiable factor that exacerbates both HFpEF and AF by promoting systemic inflammation, left atrial remodelling, and neurohormonal activation (6,7). The management of this patient was appropriately focused on
controlling heart rate, optimizing blood pressure, managing volume status, and preventing thromboembolic events. Beta-blocker therapy (metoprolol) was effective for rate control, while diuretics addressed the peripheral
edema. Losartan and hydrochlorothiazide provided additional blood pressure control and diuresis. Given the patient’s CHA2DS2-VASc score—elevated due to age, hypertension, and heart failure—anticoagulation with
apixaban was warranted to reduce the risk of stroke, which is fivefold higher in individuals with AF (5,8). Despite more than a year of rate control and anticoagulation, the patient did not experience spontaneous
reversion to sinus rhythm. Therefore, elective cardioversion was pursued, preceded by transesophageal echocardiography to rule out intracardiac thrombus. The successful restoration of sinus rhythm improved cardiac
efficiency and potentially reduced the risk of HF progression (9). Ongoing rhythm surveillance is essential, as recurrence of AF is common, particularly in patients with underlying structural heart disease and obesity (10).
From a preventive perspective, aggressive risk factor modification remains a cornerstone of long -term management. Weight reduction, dietary sodium restriction, and increased physical activity can improve diastolic
function and reduce AF recurrence (1,2). Additionally, lipid lowering with statin therapy is indicated given the elevated LDL cholesterol level (149 mg/dL), further reducing cardiovascular risk (3,6).
In summary, this case underscores the importance of a multidisciplinary approach in managing AF and HFpEF , particularly in patients with multiple cardiovascular risk factors. Optimal control of hypertension, obesity, and
dyslipidemia, combined with rhythm management and anticoagulation, can lead to favorable clinical outcomes. The patient’s successful cardioversion and stable follow-up course demonstrate that with comprehensive care,
symptom control and maintenance of sinus rhythm are achievable in this complex patient population.
References:
- Kittleson MM, Panjrath GS, Amancherla K, et al. 2023 ACC Expert Consensus Decision
Pathway on Management of Heart Failure With Preserved Ejection Fraction. J Am Coll Cardiol.
2023;81(18):1835–78. doi:10.1016/j.jacc.2023.03.393 - American College of Cardiology. 2024 ACC Expert Consensus Decision Pathway on
Clinical Assessment, Management, and Trajectory of Patients Hospitalized With Heart Failure. J Am Coll
Cardiol. 2024. doi:10.1016/j.jacc.2024.06.002 - American College of Cardiology/American Heart Association. 2024 Update to the 2020
ACC/AHA Clinical Performance and Quality Measures for Adults With Heart Failure. Circ Heart Fail/HCQ. - doi:10.1161/HCQ.0000000000000132
- Riccardi M, et al. Current Treatment of Heart Failure with Preserved Ejection Fraction. J
Clin Med. 2025;14(15):5406. doi:10.3390/jcm14155406 - European Society of Cardiology. 2024 ESC Guidelines for the Management of Atrial
Fibrillation. Eur Heart J. 2024. Available from:
https://www.escardio.org/Guidelines/Clinical-Practice-Guidelines/Atrial-Fibrillation - Shahid M, et al. A Contemporary Review on Heart Failure with Preserved Ejection
Fraction. Front Cardiovasc Med. 2024. Available from:
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC12060923/ - Romanò M, et al. Heart Failure Syndromes: Different Definitions of Ejection Fraction and
Implications. J Clin Med. 2025;14(14):5090. doi:10.3390/jcm14145090 - Spotlight on the 2024 ESC/EACTS Management of Atrial Fibrillation. Eur Heart J
Cardiovasc Pharmacother. 2025. doi:10.1093/ehjcvp/pvad014 - Drug Therapy for Acute and Chronic Heart Failure with Preserved Ejection Fraction.
PubMed Central (PMC). 2025. Available from:
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC11093799/ - Hoevelmann J. What’s New in Heart Failure? PubMed Central (PMC). 2025 Sep. Available
from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC12502451/
Clinical Student’s Companion
In 1977 I wrote up a small booklet called CLINICAL STUDENTS COMPANION and had it published by AzamSons, a book seller across from Dow Medical College. It was basically notes on General Medicine I made for the Final Professional MBBS Exam. It was common for oral examiners to ask for causes of symptoms, signs or abnormal lab values. This helped junior students to brush up on lists of causes which one could not find easily from text books of Medicine like Davidson or Harrison or Cecil Loeb. I then published two more editions wht last one being what is being uploaded here, a 260 page book with illustrations. The third edition was typed by me on the old manual Smith Corona portable type-writer.
One technique we learnt from teachers like Prof. S.M Rab (with whom I did my first intership in Medicine at Jinnah PostGraduate Medical Center, Karachi) was to think about the causes of a presentation or symptom or sign one at a time. If patient has 2-4 signs think of all the causes separately and try to figure what runs common to all the lists. This may lead one to correct diagnosis. For example when presented with Fever of Unknown Origin, think of the causes and if the patient also has anemia and splinter hemorrhages, one would find that bacterial endocarditis is present in all the 3 lists. Same for other problems.
Hope you enjoy reading. Mind you this is 45 years ago and applies to diseases found commonly in Pakistan and India. It’s value at this stage may be more historical than actual since everything now is a mouse click away on ChatGPT! 🙂 Amin H. Karim MD September 25 2025
Circ Under Duress
A CASE OF POSTOPERATIVE COMPLICATION OF SURGICAL MITRAL VALVE REPLACEMENT.
Amin H. Karim MD
CASE:
A 71 year olf Caucasian male with history of hypertension and hyperlipidemia, and chronic atrial fibrillation, developed non-rheumatic mitral valve regurgitation secondary to mitral valve prolapse, which had gradually progressed over a decade years to severe regurgitation, accompanied by moderately severe tricuspid regurgitation. Patient tolerated the valvular lesions for many years till he became symptomatic with NYHA Class III dyspnea and agreed for intervention.
CARDIAC MRI: In November 2024, cardiac MRI revealed bi-leaflet mitral valve prolapse, with severe left atrial enlargement and moderate tricuspid regurgitation. The global ejection fraction was 65% with biventricular dilatation, and basal and mid inferolateral wall scarring. Mitral and tricuspid annulus were dilated.
TWO DIMENSIONAL ECHOCARDIOGRAM: On Echocardiogram, right ventricular function was low normal. Left ventricular ejection fraction was normal. There was mitral valve prolapse due to myxomatous degeneration, severe mitral regurgitation with regurgitation fraction of 51% and estimated regurgitant volume of 56 ml. Pulmonary artery pressures were normal. Cardiac catheterization showed normal coronaries
With a low BMI and good overall health, he was felt by the cardiovascular surgeon to be low risk for surgery and MitraClip therefore not warranted.
MITRAL VALVE REPAIR SURGERY:
Mitral valve repair with a 34 mm physio 2 flexible annuloplasty ring; Intra-aortic balloon pump; Tricuspid valve repair with a 28 mm triad rigid ring; Biatrial maze utilizing the encompass clamp, RF clamp and cryoprobe; Left atrial appendage ligation with a 45 mm atrial cure mini atrial clip:
Following mitral valve repair there was no mitral regurgitation with a long segment of coaptation beneath the annular plane. The transmitral gradient was 1 mm Hg. Similarly following tricuspid valve repair there was no regurgitation with the trans tricuspid valve gradient of 1 mm.
Following bi-atrial maze procedure, the patient converted to sinus rhythm. However, when the cardio pulmonary bypass was reversed and the patient was being closed, he went to ventricular fibrillation followed by defibrillation. Frequent PVCs were observed. Intravenous amiodarone and magnesium were started; His blood pressure started to drop and vasopressors were increased and intra-aortic balloon placed. His hemodynamics improved and was watched in the OR with echo. There was inferior wall hypokinesis. He was maintained on low dose dobutamine, vasopressin and norepinephrine. His global ejection fraction was reasonable; chest was closed, but before he could be transferred out of OR he developed ventricular fibrillation again. His chest was opened and direct cardiac massage and cardioversion done with return of circulation. ECMO (Extracorporeal Membrane Oxygenation) was initiated. He was transferred to the cardiac cath lab and underwent emergency coronary angiography.
CORONARY INTERVENTION: Coronary angiography showed the dominant left circumflex was occluded in the mid potion.
IVUS showed possible edema around the left circumflex and it was felt that the circumflex was occluded due to pressure from the mitral valve ring. Stent was placed with recanalization. Impella was placed in the cath lab and the intra-aortic balloon was removed.
Intavascular Ultrasound (IVUS) shows the edema/hematoma? aeound the left vircumflex artery.
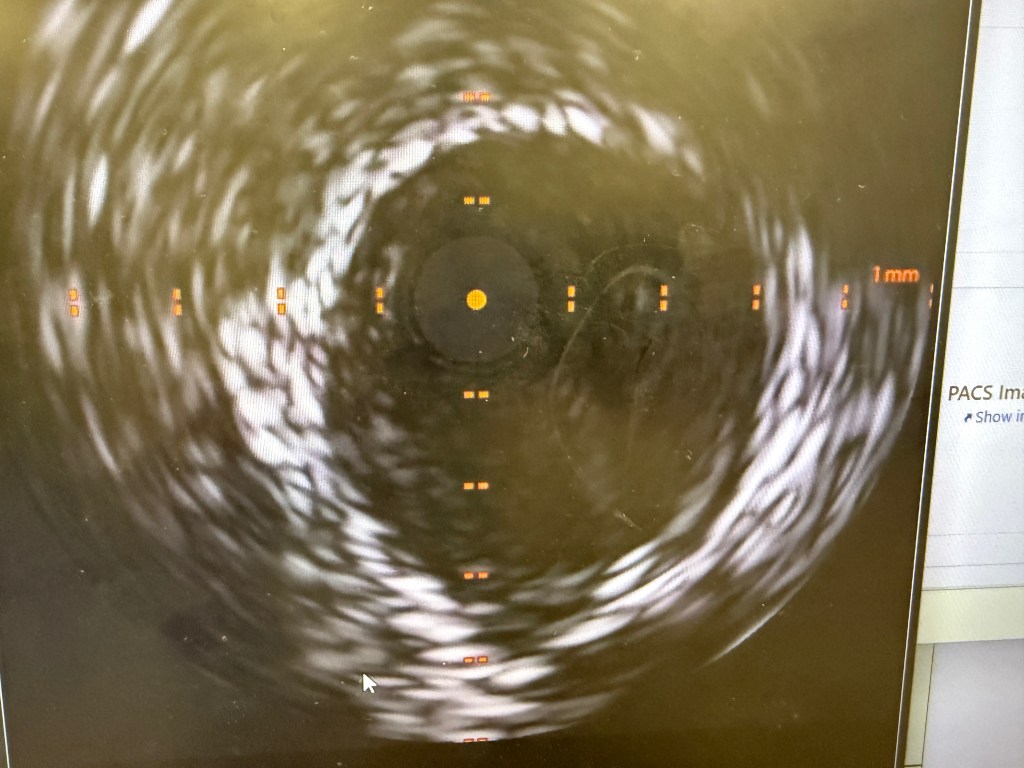
A 3.5 mm x 32 mm Synergy drug eluting stent was placed in the mid circumflex coronary artery with a good result. The intra-aortic balloon was removed.
INTENSIVE CARE UNIT: Patient remained with supported blood pressure, with severe anemia needing multiple blood transfusions. Transesophageal echo showed severely depressed right and left ventricular systolic function.
Patient was taken back to O.R. for exploration and washout. On return to ICU noted to have ST elevation in lead VII. Taken back to cath lab and coronary angiography done showing that the circumflex stent was patent.
FOLLOWUP: 10 days after mitral and tricuspid valve repair and coronary intervention, patient is off ECMO, and on Impella support. He is awake but does not follow commands. His global ejection fraction on echocardiogram is mildly depressed (40-45%) with trace of mitral and tricuspid regurgitation.
The Case of Asymptomatic Aortic Stenosis: Lessons in Patient Education and Early Symptom Recognition
Authors:
Ana Maria Sierra Valiente
Universidad Anahuac Mexico Norte
Joy Emem Efik, MBBS
Sumy State University
Ayesha Asghar, MBBS
Multan Medical and Dental College, Pakistan.
Sarai Anayansi Zárate Chavez
Universidad Anahuac Oaxaca
Amin H. Karim MD, FRCP
Baylor College of Medicine, Houston
Academy of Medicine, Houston
Weill Medical College of Cornell University.
The Skin-Heart Connection
By
Ana María Sierra Valiente Universidad Anáhuac México Norte
Amin H. Karim MD,
Clinical Associate Professor
Baylor College of Medicine, Houston, Texas.
Interrupting Anticoagulants for Surgery
Interrupting Anticoagulants for Surgery: Guidelines, Risk Stratification, and Clinical Decision-Making.
Raya Kharboutli PA-S2
University of Texas Medical Branch

Regina Medina Urrutia
Universidad Anahuac Campus Xalapa

Anticoagulants are a class of medications used to prevent and treat thromboembolic events such as stroke, deep vein thrombosis, and pulmonary embolism. These agents are divided into two main categories: antiplatelet agents and anticoagulants that inhibit coagulation factors. Understanding their mechanisms of action is critical for making safe perioperative decisions.
Antiplatelet agents like aspirin irreversibly inhibit cyclooxygenase (COX-1), preventing the production of thromboxane A₂, a molecule essential for platelet activation. As a result, platelet aggregation is impaired for the lifespan of the platelet, which is approximately 7-9 days (1). Clopidogrel, a P2Y12 receptor inhibitor, irreversibly blocks ADP receptors on the platelet surface, further preventing platelet activation and aggregation. Following discontinuation, platelet function typically returns to baseline within about 5 days (2). Warfarin is a vitamin K antagonist that works by inhibiting vitamin K epoxide reductase, an enzyme required for the activation of clotting factors II, VII, IX, and X. Warfarin has a delayed onset of action, with therapeutic anticoagulation typically achieved within 2 to 3 days. Monitoring is performed using International Normalized Ratio (INR), and full reversal of anticoagulant effect takes approximately 3 to 5 days. This process can be expedited with the administration of vitamin K (3). Direct oral anticoagulants (DOACs) are a newer class of medications with more predictable pharmacokinetics. These include factor Xa inhibitors such as apixaban, rivaroxaban, and edoxaban, which inhibit factor Xa, thereby blocking the conversion of prothrombin to thrombin. Additionally, dabigatran is a direct thrombin inhibitor (Factor IIa), which prevents the conversion of fibrinogen to fibrin, the final step in clot formation (4).
The type of anticoagulant used plays an important role in how far in advance it should be discontinued:
- Warfarin should usually be stopped 5 days before surgery to allow the INR to return to a safe range (usually <1.5) (11).
- Direct oral anticoagulants (DOACs) like apixaban, rivaroxaban, dabigatran, and edoxaban are usually suspended 24–72 hours before surgery, depending on the bleeding risk of the procedure and the patient’s kidney function. For example, dabigatran is mostly eliminated by the kidneys, so patients with renal impairment need to stop it even earlier (11).
This classification and understanding of mechanisms provide a foundation for evaluating how and when these agents should be temporarily discontinued prior to surgical or invasive procedures, based on the individual agent, patient thrombotic risk, and the bleeding risk associated with the procedure
Interrupting anticoagulation before a procedure is often necessary to reduce the risk of excessive bleeding during or after surgery. Anticoagulants and antiplatelet agents impair the body’s ability to form clots, which is beneficial for preventing thrombosis but can lead to significant complications when tissue trauma or vascular injury is expected. The decision to pause these medications is a balance between two major risks: bleeding and thrombosis. For procedures with a high bleeding risk, such as major surgeries, spinal or epidural anesthesia, and certain endoscopic or urologic procedures, continued anticoagulation can increase the chance of uncontrolled bleeding, hematoma formation, or the need for transfusions (5). On the other hand, abruptly stopping antithrombotic therapy, especially in high-risk patients (such as those with recent stroke, atrial fibrillation, or coronary stents), may raise the risk of life-threatening thromboembolic events (7). Therefore, clinicians must evaluate the type of anticoagulant, the patient’s thrombotic risk, and the bleeding risk of the procedure to determine the safest perioperative plan. In many cases, temporary interruption with or without bridging therapy allows for safe procedural outcomes while minimizing harm from both bleeding and clot formation (6).
In cardiology, several invasive procedures carry moderate to high bleeding risk and typically require temporary interruption of anticoagulant or antiplatelet therapy. The decision depends on the type of medication, the procedure’s bleeding risk, and the patient’s thromboembolic risk. For cardiac surgery, such as coronary artery bypass grafting (CABG) or valve replacement, both antiplatelet agents and anticoagulants are usually interrupted. Aspirin is often continued unless bleeding risk is very high but clopidogrel is typically discontinued at least 5-7 days before surgery to minimize perioperative bleeding (8). Warfarin is usually stopped 5 days prior, aiming for an INR of less than 1.5 on the day of the surgery. In patients at high thromboembolic risk such as mechanical valve or atrial fibrillation with prior stroke, bridging with low molecular weight heparin (LMWH) may be considered. Direct oral anticoagulants are typically held for 2-3 days before major cardiac surgery, with the exact timing depending on renal function.
For pacemaker or implantable cardioverter-defibrillator (ICD) insertion, the bleeding risk is considered moderate. Aspirin may be continued in most cases, but clopidogrel should be stopped 5-7 days prior, especially if dual antiplatelet therapy is not mandatory at the time. DOACs are commonly interrupted 24-48 hours before the procedure, depending on renal function and Warfarin is often continued at a therapeutic INR for minor device procedures, but only interrupted in high-bleeding-risk cases (9). Percutaneous coronary intervention (PCI) presents a unique challenge, especially in patients already on dual antiplatelet therapy (DAPT). These procedures are rarely elective if DAPT is indicated. If non-urgent PCI must be delayed, clopidogrel is held 5-7 days and DOACs for 48-72 hours prior (10).
Ultimately, the goal is to minimize both bleeding and thrombotic complications by tailoring medication interruption based on the procedure type, medication half-life, and patient risk factors.
The management of patients going under anesthesia for surgery is a really common challenge due to the decision to suspend or not the anticoagulants the patients are on. Many protocols can be followed to help make the decision. One of these protocols is to evaluate both the risk of bleeding and thromboembolism, and it’s important to know the dosage of the anticoagulant and the reasons why the patient is taking the specific anticoagulant.
First of all, the risk of bleeding needs to be estimated. One way is the HAS-BLEED score, which will assess the following risk factors such as hypertension, abnormal liver or renal functions, stroke, bleeding, labile INRs, elderly patients (>65 years), and the use of drugs or alcohol. The second step is to estimate the thromboembolic risk, and to do that, age and comorbidities need to be evaluated (12). If the patient has had a recent event of DVT or PE, the decision is based on the diagnosis, but in this scenario, the surgery is delayed as much as possible. Once the two important risks are evaluated, the duration to interrupt the anticoagulant is going to depend on which medication the patient is on. If the patient has low kidney or liver function, we might need additional consideration. In general, almost every procedure, the anticoagulant must be suspended if the risk of bleeding or high thrombotic risk, but if the risk is low isn’t necessarily necessary to stop the medication (12).
There are really selected procedures where we can keep using the anticoagulant, like in a dental extraction, skin biopsy, or a cataract surgery, but also in a procedure like a cardiac implant electronic device, it’s not necessary to stop taking them. The ERHA states that if the patient is going to be under the implantation of a cardiac electronic device like a pacemaker, the patient should continue the anticoagulant perioperatively (13). Unless the patient has a risk of a thromboembolic event and is under warfarin or DOCAs, the medication should be suspended temporarily. In the case of any endovascular procedures like an angioplasty, a meta-analysis randomly shows that patients who were under warfarin and didn’t interrupt while undergoing the procedure were associated with lower risks of complications compared with those who interrupted the warfarin perioperatively (11).
In patients with high thrombotic risk, it may be necessary to use bridging therapy with low-molecular-weight heparin (LMWH) during the time the oral anticoagulant is stopped. However, the BRIDGE trial showed that bridging in patients with non-valvular atrial fibrillation and moderate thrombotic risk increased the risk of bleeding without significantly reducing thromboembolic events (14). Therefore, bridging should only be considered in selected high-risk patients.
When using spinal or epidural anesthesia, anticoagulants increase the risk of spinal hematoma, which can cause permanent paralysis. According to the American Society of Regional Anesthesia (ASRA), anticoagulants such as DOACs should be stopped at least 72 hours before any neuraxial procedures, and specific guidelines should be followed for restarting the medication (15).
Individual characteristics such as renal or liver function, age, history of bleeding, and the use of other medications like antiplatelet agents or NSAIDs, must also be considered when deciding whether to stop anticoagulants before surgery (11). Restarting anticoagulants too soon can lead to postoperative bleeding, while delaying them too long can cause thromboembolism. In general, anticoagulants can be restarted 24–48 hours after surgery if bleeding is under control and the patient is stable (11).
References:
- Haut, E. R., Pronovost, P. J., & Owings, J. T. (2016). Thromboembolic complications in trauma patients. Trauma Surgery & Acute Care Open, 1(1), e000022. https://doi.org/10.1136/tsaco-2015-000022
- Pannucci, C. J., & Dresher, M. (2017). Postoperative venous thromboembolism: Risk factors and prevention. International Journal of Environmental Research and Public Health, 14(3), 301. https://doi.org/10.3390/ijerph14030301
- Hirsh, J., Guyatt, G., Albers, G. W., Harrington, R., & Schünemann, H. J. (2008). Antithrombotic and thrombolytic therapy: American College of Chest Physicians evidence-based clinical practice guidelines (8th edition). Circulation, 107, 1692–1700. https://doi.org/10.1161/01.CIR.0000063575.17904.4E
- Zareba, W. (2020). Perioperative management of patients receiving anticoagulants. In StatPearls. StatPearls Publishing. https://www.ncbi.nlm.nih.gov/books/NBK557590/
- Guyatt, G. H., Akl, E. A., Hirsh, J., Crowther, M., Gutterman, D. D., & Schünemann, H. J. (2022). American College of Chest Physicians Antithrombotic Guidelines. Chest, 161(5), 1272–1302. https://doi.org/10.1016/j.chest.2022.01.032
- Nasser, M., Jaffer, A. K., Milani, R. V., & Lavie, C. J. (2021). Perioperative management of anticoagulants in patients undergoing elective procedures. Perioperative Medicine, 10(1), 1–9. https://doi.org/10.1186/s13741-020-00170-4
- National Library of Medicine. (n.d.). Bethesda Statement on Open Access Publishing. PubMed. https://pubmed.ncbi.nlm.nih.gov/static-page/down_bethesda.html
- Kakar, T. S., Elbaroni, M., & Kang, N. (2020). Bridging anticoagulation therapy in patients undergoing procedures: A literature review. Cardiology Research, 11(5), 328–334. https://doi.org/10.14740/cr1110
- Douketis, J. D., Spyropoulos, A. C., Duncan, J., Carrier, M., Le Gal, G., & Tafur, A. J. (2021). Perioperative management of anticoagulant and antiplatelet therapy. Thrombosis Journal, 19(1), 29. https://doi.org/10.1186/s12959-021-00279-6
- Douketis, J. D., Spyropoulos, A. C., Kaatz, S., Becker, R. C., Caprini, J. A., Dunn, A. S., … & Schulman, S. (2015). Perioperative bridging anticoagulation in patients with atrial fibrillation. New England Journal of Medicine, 373(9), 823–833. https://doi.org/10.1056/NEJMoa1302946
- Kaicker, J., Mokrzycki, M. H., & Salvador, D. (2024). Bleeding risk assessment and anticoagulant management during surgery. PubMed. https://pubmed.ncbi.nlm.nih.gov/38320132/
- UpToDate. (2024). Perioperative management of patients receiving anticoagulants. UpToDate. https://www.uptodate.com/contents/perioperative-management-of-patients-receiving-anticoagulants
- UpToDate. (2024). Periprocedural management of antithrombotic therapy in patients receiving long-term oral anticoagulation and undergoing percutaneous coronary intervention. UpToDate. https://www.uptodate.com/contents/periprocedural-management-of-antithrombotic-therapy-in-patients-receiving-long-term-oral-anticoagulation-and-undergoing-percutaneous-coronary-intervention/print
- Heidbuchel, H., Verhamme, P., Alings, M., Antz, M., Hacke, W., Oldgren, J., … & Lip, G. Y. H. (2015). Updated European Heart Rhythm Association practical guide on the use of non-vitamin K antagonist anticoagulants in patients with non-valvular atrial fibrillation. Europace, 17(10), 1467–1507. https://doi.org/10.1093/europace/euv309
- Macedo, A. F., Bell, J., McCarron, C., & Fahey, T. (2018). Interventions to improve appropriate prescribing of anticoagulants in atrial fibrillation: A systematic review. BMC Cardiovascular Disorders, 18(1), 24. https://doi.org/10.1186/s12872-018-0762-1
Intracardiac Clots: From Formation to Clinical Management.
Sarai Anayansi Zárate Chávez
Universidad Anáhuac campus Oaxaca

Juan Pablo García Guzmán
Universidad Anáhuac Mexico campus Norte

Amin H. Karim MD
Institute of Academic Medicine, Houston, Texas
Weill Medical College of Cornell University.
What Is a Blood Clot?
A blood clot also referred to as a thrombus (plural: thrombi), intravascular clot, or coagulum is a gelatinous or semi-solid mass of coagulated blood that forms within the circulatory system. When such a clot develops in the deep venous system, most commonly in the lower limbs, it is termed deep vein thrombosis (DVT), although it can also occur in the upper extremities.
A major complication of DVT is embolization, in which one or more thrombi detach and travel through the venous circulation often originating in the legs, pelvis, or groin and reach the pulmonary arteries, leading to a pulmonary embolism (PE). This condition can be life-threatening and requires immediate medical intervention.
Thrombus Formation and Intracardiac Clot Dynamics
A thrombus also referred to as a clot, blood clot, embolus (when mobile), or
thromboembolus (when causing obstruction) is the result of a complex interaction between endothelial injury, abnormal blood flow (stasis or turbulence), and a hypercoagulable state, often summarized by Virchow’s triad.
In the setting of acute vascular injury, particularly in acute coronary syndrome
(ACS), clot formation begins with platelet adhesion to exposed subendothelial
proteins at sites of plaque rupture or erosion. Once adhered, platelets become
activated, change shape, and release a variety of pro-thrombotic substances
including thromboxane A2, ADP, and serotonin, promoting further platelet
activation and local vasoconstriction. The surface expression of glycoprotein IIb/IIIa receptors increases, facilitating platelet aggregation through fibrinogen bridging. Concurrently, the coagulation cascade is triggered, leading to thrombin generation. Thrombin amplifies platelet activation and converts fibrinogen into fibrin, which stabilizes the growing thrombus. As fibrin is laid down, a stable platelet-fibrin thrombus forms, which may partially or completely obstruct the vessel. If embolized, fragments of the thrombus may lodge downstream, causing ischemia or infarction.
Intracardiac thrombi form under somewhat different circumstances, often related to blood stasis or structural heart disease. In the left ventricle, thrombi can arise after anterior myocardial infarction, especially with regional wall motion abnormalities such as apical akinesis or dyskinesis. In non-ischemic dilated cardiomyopathy, the risk is lower but still present, particularly when left ventricular ejection fraction is severely reduced.
The left atrium, particularly the left atrial appendage, is a common site for thrombus formation in patients with atrial fibrillation, atrial flutter, or significant mitral valve disease. Even in sinus rhythm, atrial mechanical dysfunction—as in cardiac amyloidosis—can predispose to thrombus formation. On the right side of the heart, thrombi may form in cases of central venous catheters, intracardiac devices, severe right ventricular dysfunction, or
hypercoagulable states. Additionally, mechanical prosthetic valves, especially with inadequate anticoagulation, are a high-risk source of thrombus formation and systemic embolism. Paradoxical embolism can occur in the presence of a patent foramen ovale (PFO) or atrial septal defect (ASD), where venous thrombi bypass the pulmonary circulation and enter the systemic arterial system through a right-to-left intracardiac shunt.
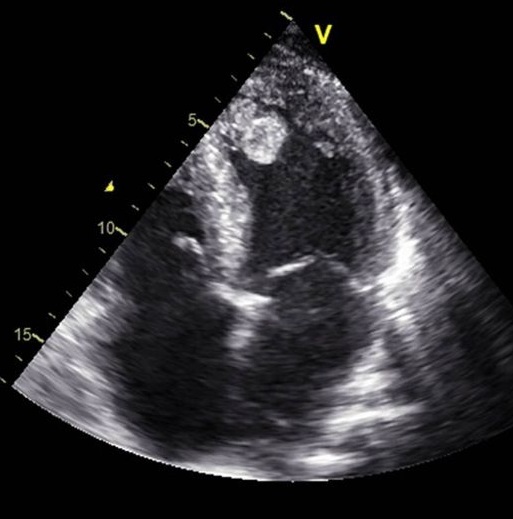
Diagnosis: Tests
The main diagnostic tests for detecting thrombi in the left ventricle are transthoracic echocardiography (TTE) and cardiac magnetic resonance imaging (CMRI) with delayed gadolinium enhancement. TTE is the most used initial technique due to its availability and low cost: however, its sensitivity is limited (approximately 21-35%), although its specificity is high (95-98%). The use of intravenous contrast agents in TTE improves sensitivity (up to 64%) without losing specificity. Transthoracic echocardiography has been utilized for identifying left ventricular thrombi since the early 1980s. In recent years, the introduction of echocardiographic contrast agents has improved detections accuracy, particularly in patients with suboptimal acoustic windows. TTE remains the initial diagnostic modality of choice for evaluating left ventricular thrombus. However, its limitations such as difficulty imaging patients with poor acoustic windows, can lead to considerable interobserver variability, potentially compromising diagnostic reliability. Cardiac magnetic resonance offers a diagnostic edge over echocardiography by allowing both myocardial tissue characterization and dynamic imaging. With recent advancements in imaging sequences and the use of paramagnetic contrast agents to enhance blood pool visualization, late gadolinium enhancement CMR may offer superior sensitivity for detecting left ventricular thrombi.
Recent epidemiologic tests have provided that the incidence of left ventricular
thrombus, using optimal imaging modalities, can reach up to 15% in patients with ST segment elevation myocardial infarction and up to 25% in those with anterior myocardial infarction. Although a standard transthoracic echocardiogram is frequently used for initial screening, its low sensitivity in detecting left ventricular thrombus requires the use of contrast (when not contraindicated) and/or cardiac MRI when there is a high pretest probability.
Transesophageal echocardiography does not provide advantages for visualizing
the ventricular apex and is not recommended as a second-line method for
ventricular thrombi.
The first study that was able to compare the diagnostic accuracies of CMRI,
contrast TTE and noncontrast TTE was performed by Weinsaft et al. That
demonstrated that even with administration of echocardiographic contrast agents, CMRI was still considerably more accurate modality in terms of thrombus detection.
CMR with late gadolinium enhancement is the gold standard, with a sensitivity of 82-88% and specificity of 99-100%, as it allows differentiation of the thrombus (avascular without enhancement) from the surrounding myocardium. It is especially recommended when TTE (even with contrast) is not diagnostic or clinical suspicion persists. Cardiac computed tomography can incidentally detect thrombi, but it is not validated for this purpose
Precise detection of left ventricular thrombi is crucial, as it frequently guides the initiation of anticoagulation therapy to reduce the risk of embolic complications. While current guidelines suggest that starting anticoagulation may be reasonable in patients with strong suspicion of thrombus such as those with apical akinesis or dyskinesis even without visible thrombus, selecting the most appropriate imaging modality is essential to ensure timely and evidence-based therapeutic decisions.
Complications
The main complications of thrombi in the left ventricle are systemic embolic events, especially ischemic stroke and peripheral arterial embolisms. Embolization occurs because the thrombus can detach and migrate into systemic circulation, affecting organs such as the brain, kidneys, spleen, or extremities. The risk of embolization is particularly high in the first few weeks after an acute myocardial infarction and can reach up to 22% depending on the morphology and follow up of the thrombus.
The incidence of systemic embolic events in patients with left ventricular thrombi varies depending on the population and clinical context. In patients with acute myocardial infarction (AMI), the incidence of left ventricular thrombus is 3.5% to 7.1% after previous AMI when cardiac magnetic resonance imaging is used, and the incidence of systemic embolism (including stroke) in the presence of thrombus is between 7% and 16% in the first few years after the event, with an annualized risk of 3.7% compared to 0.8% in patients without left ventricular thrombus. Other relevant complications include major adverse cardiovascular events (MACE), which include death, reinfarction, and hospitalization for heart failure. The presence of left ventricular thrombus is associated with a significant increase in mortality and long-term adverse cardiovascular events. In addition, patients with persistent thrombus are at increased risk of bleeding, especially if they require prolonged anticoagulation.
The American Heart Association emphasizes that complete thrombus resolution is associated with lower mortality, while thrombus persistence, especially if mural and organized, carries a lower but not zero risk of embolization. The patient groups with the highest incidence of complications associated with thrombi in the left ventricle are mainly those with extensive acute myocardial infarction (AMI), especially anterior AMI, patients with ventricular aneurysm, and those with reduced left ventricular ejection fraction (LVEF). In addition, patients with dilated cardiomyopathy, either ischemic or non-ischemic, particularly those with severe systolic dysfunction, also have an elevated risk of embolic complications and major cardiovascular events.
In the context of non-ischemic cardiomyopathy, patients with dilated
cardiomyopathy show an even higher risk of systemic embolism compared to other non-ischemic etiologies and ischemic heart disease. The presence of mobile or protruding thrombi increases the risk of embolization, while thrombus persistence is associated with higher mortality and adverse events.
The American Heart Association points out that the combination of anterior AMI, low LVEF, ventricular aneurysm, and delayed reperfusion are factors that identify patients at higher risk of embolic complications and mortality associated with thrombi in the left ventricle.
The factors that increase the risk of thrombus formation in the left ventricle vary depending on the patient group, but they share pathophysiological mechanisms based on Virchow’s triad: ventricular dysfunction (stasis), endocardial damage, and inflammation/hypercoagulability. In patients with extensive acute myocardial infarction (AMI), especially anterior AMI, the highest risk factors are anterior location of the infarction, presence of ventricular aneurysm, left ventricular ejection fraction (LVEF) <30-40%, larger infarction size (elevated troponins), delayed reperfusion, and suboptimal coronary flow after intervention. The combination of reduced LVEF and segmental dysfunction (particularly apical) is the main predictor of thrombus and embolic complications or major cardiovascular events in all these groups. Systemic inflammation (elevated CRP) and the use of certain antithrombotic drugs may also contribute
Managment
Management of left Heart Thrombi (RHT)
The cornrstone of managment for intracardiac thrombus, particularly left ventricular thrombus, is therapeutic anticoagulantion. This strategy aims to reduce the risk of systemic embolism and promote trhombus resolution. Anticoagulation should be initiated promptly upon diagnosis, typically with intravenous unfractionated heparin, low molecular weight heparin, or a direct oral anticoagulant (DOAC). Transition to oral therapy with either warfarin or a DOAC is the recommended available evidence suggests that anticoagulation significantly lowers embolic risk and increases the likelihood of thrombus resolution compared to no or subtherapeutic treatment. In particular, a higher time in therapeutic range with warfarin is associated with superior outcomes and appears to outweigh the bleeding risks, even in the presence of concurrent antiplatelet therapy. The standard duration of anticoagulation is a minimum of three months.
Follow-up
cardiac imaging, ideally using the same modality employed at diagnosis, should be performed at that point to assess thrombus resolution. If the thrombus persists without notable change, anticoagulation should be continued with periodic reassessment. In cases where the thrombus has decreased in size or displays features consistent with chronicity and reduced embolic potential, the decision to continue therapy should be based on ongoing embolic risk, such as persistent left ventricular dysfunction, aneurysm formation, or spontaneous echocardiographic contrast. If both the thrombus and contributing risk factors have resolved, evidenced by normalization of systolic function and absence of additional indications for anticoagulation, discontinuation of therapy may be appropriate. For patients who develop LVT in the context of prior MI (≥3 months) or chronic ischemic cardiomyopathy, no randomized controlled data exist to guide treatment duration. Nonetheless, anticoagulation for a period of 3 to 6 months is generally recommended. Beyond that, extended or indefinite therapy should be considered on a case-by-case basis, incorporating individual thrombotic and bleeding risks, recovery of ventricular function, and patient preferences through shared decision-
making.
Management of Right Heart Thrombi (RHT)
Right heart thrombi (RHT) are rare but potentially life-threatening findings, often associated with pulmonary embolism (PE) and right ventricular dysfunction. The management of RHT remains a clinical challenge due to the lack of randomized controlled trials and standardized treatment guidelines. However, observational studies and registry data suggest that anticoagulation alone is often insufficient, especially in cases involving mobile or serpiginous thrombi with high embolic potential.
Initial management typically includes systemic anticoagulation with intravenous unfractionated heparin or low molecular weight heparin. This serves as a bridge to definitive therapy and may be appropriate in hemodynamically stable patients with non-mobile thrombi or contraindications to more aggressive interventions.
For patients with mobile RHT or hemodynamic compromise, reperfusion strategies are generally preferred. Systemic thrombolysis has demonstrated lower mortality rates compared to anticoagulation alone, but carries a notable risk of major bleeding, including intracranial hemorrhage. Surgical embolectomy is another option, particularly in patients with contraindications to thrombolysis or when thrombi are large, organized, or entangled in cardiac structures.
Catheter-directed therapies, including percutaneous aspiration thrombectomy (e.g., AngioVac, FlowTriever, AlphaVac), have gained attention as minimally invasive alternatives. These techniques allow for rapid thrombus removal with high success rates and a lower bleeding profile compared to systemic thrombolysis. Early outcomes are promising, although data remain limited and long-term efficacy has not been firmly established.
Ultimately, the choice of therapy should be guided by thrombus characteristics
(size, mobility, morphology), patient stability, comorbidities, bleeding risk, and institutional expertise. In general, mobile RHTs or those associated with acute PE warrant urgent intervention beyond anticoagulation alone. Multidisciplinary decision making often involving cardiology, critical care, interventional radiology, and cardiothoracic surgery is essential for optimizing outcomes.
Prevention
Intracardiac thrombus formation is a recognized complication in patients with heart failure and reduced ejection fraction, particularly in those with non-ischemic dilated
cardiomyopathy (DCM). Although left ventricular (LV) thrombi are more frequently documented, thrombi may also develop in the right heart chambers, especially in the presence of right-sided dysfunction, central venous catheters, cardiac devices, or systemic hypercoagulable states.
The use of antithrombotic therapy for primary prevention of thrombus formation in this population remains a subject of ongoing clinical judgment. In patients with DCM who are in sinus rhythm and without prior thromboembolic events, neither aspirin nor warfarin has consistently demonstrated clear benefit in preventing thrombus formation or reducing major adverse cardiovascular events. Therefore, routine prophylactic use of these agents is generally not recommended. However, individualized assessment is essential, especially when additional risk factors such as atrial fibrillation, prior embolic events, severely reduced ejection fraction, or left ventricular aneurysms are present.
In select subtypes of DCM that carry a higher inherent risk of intracardiac thrombus such as Takotsubo syndrome with apical ballooning, left ventricular
noncompaction, peripartum cardiomyopathy, eosinophilic myocarditis, and
infiltrative diseases like cardiac amyloidosis the use of oral anticoagulants (e.g.,
warfarin) or parenteral agents may be considered on a case-by-case basis. In
contrast, low-dose aspirin may offer some theoretical antiplatelet benefit, but its role in thrombus prevention remains less defined. Long-term anticoagulation may be appropriate for patients with persistent ventricular dysfunction or recurrent thromboembolic risk, provided the bleeding risk is acceptable.
Bibliografia link
Mathevosian, S., & Ranade, M. (2022). Right Heart Clot-in-Transit: Endovascular
Therapies. Seminars in interventional radiology, 39(5), 515–522.
https://doi.org/10.1055/s-0042-1757942
Sakellariou, X. M., Efstathopoulos, A., Stamatis, K. V., Nikas, D. N., & Kolettis, T.
M. (2020). Treatment of Mobile Right Heart Thrombi. European journal of case
reports in internal medicine, 7(12), 001918. https://doi.org/10.12890/2020_001918
Patel, A. N., Amrutiya, R. J., Manvar, B. N., & Patel, A. (2022). A proposed
approach for the management of clot-in-transit. Cureus, 14(8).
Lawrence LK Leung, M. D. (2025). Overview of hemostasis.
https://shorturl.at/hkxsH
Levine, G. N., McEvoy, J. W., Fang, J. C., Ibeh, C., McCarthy, C. P., Misra, A.,
Shah, Z. I., Shenoy, C., Spinler, S. A., Vallurupalli, S., Lip, G. Y. H., on behalf of
the American Heart Association Council on Clinical Cardiology, Council on
Cardiovascular and Stroke Nursing, & and, S. C. (2022). Management of patients at
risk for and with left ventricular thrombus: A scientific statement from the american
heart association. Circulation, 146(15), e205–e223.
10.1161/CIR.0000000000001092
Warren J Manning, M. D. (2024). Echocardiography in detection of cardiac and
aortic sources of systemic embolism. https://shorturl.at/mR7gN
Watson, N. W., Weinberg, I., Dicks, A. B., Carroll, B. J., & Secemsky, E. A. (2024).
Clinical outcomes and predictors of advanced therapy for the management of right
heart thrombus. Circulation: Cardiovascular Interventions, 17(4), e013637.
10.1161/CIRCINTERVENTIONS.123.013637
Wilson S Colucci, M. D., & Gregory YH Lip, MD, FRCPE, FESC, FACC. (2025).
Left ventricular thrombus. https://shorturl.at/Zf9oO
Kleindorfer, D. O., Towfighi, A., Chaturvedi, S., Cockroft, K. M., Gutierrez, J.,
Lombardi-Hill, D., Kamel, H., Kernan, W. N., Kittner, S. J., Leira, E. C., Lennon,
O., Meschia, J. F., Nguyen, T. N., Pollak, P. M., Santangeli, P., Sharrief, A. Z.,
Smith, S. C., Jr, Turan, T. N., & Williams, L. S. (2021). 2021 guideline for the
prevention of stroke in patients with stroke and transient ischemic attack: A
guideline from the American heart association/American stroke association. Stroke;
a Journal of Cerebral Circulation, 52(7), e364–e467.
https://doi.org/10.1161/STR.0000000000000375
Levine, G. N., McEvoy, J. W., Fang, J. C., Ibeh, C., McCarthy, C. P., Misra, A.,
Shah, Z. I., Shenoy, C., Spinler, S. A., Vallurupalli, S., Lip, G. Y. H., & American
Heart Association Council on Clinical Cardiology; Council on Cardiovascular and
Stroke Nursing; and Stroke Council. (2022). Management of patients at risk for and
with left ventricular thrombus: A scientific statement from the American Heart
Association. Circulation, 146(15), e205–e223.
https://doi.org/10.1161/CIR.0000000000001092
Camaj, A., Fuster, V., Giustino, G., Bienstock, S. W., Sternheim, D., Mehran, R.,
Dangas, G. D., Kini, A., Sharma, S. K., Halperin, J., Dweck, M. R., & Goldman, M.
E. (2022). Left ventricular thrombus following acute myocardial infarction: JACC
state-of-the-art review. Journal of the American College of Cardiology, 79(10),
1010–1022. https://doi.org/10.1016/j.jacc.2022.01.011
Sharma, N. D., McCullough, P. A., Philbin, E. F., & Weaver, W. D. (2000). Left
ventricular thrombus and subsequent thromboembolism in patients with severe
systolic dysfunction. Chest, 117(2), 314–320.
https://doi.org/10.1378/chest.117.2.314
Ram, P., Shah, M., Sirinvaravong, N., Lo, K. B., Patil, S., Patel, B., Tripathi, B.,
Garg, L., & Figueredo, V. (2018). Left ventricular thrombosis in acute anterior
myocardial infarction: Evaluation of hospital mortality, thromboembolism, and
bleeding. Clinical Cardiology, 41(10), 1289–1296. https://doi.org/10.1002/clc.23039
Albaeni, A., Chatila, K., Beydoun, H. A., Beydoun, M. A., Morsy, M., & Khalife,
W. I. (2020). In-hospital left ventricular thrombus following ST-elevation
myocardial infarction. International Journal of Cardiology, 299, 1–6.
https://doi.org/10.1016/j.ijcard.2019.07.070